Poster Presentation Australian Diabetes Society and the Australian Diabetes Educators Association Annual Scientific Meeting 2014
Insulin sensitivity differences using surrogate markers in fat depots of high fat fed obese mice (#242)
Different fat depots vary in their sensitivity to insulin, a component of insulin resistance (IR) but few studies have compared this with the profile of adipogenic, inflammatory and fibrotic markers in the various fat depots. Therefore in this study we used qPCR to examine at the mRNA level expression of these markers in adipose tissue from subcutaneous (SAT), epididymal (EAT) and brown (BAT) fat in mice.
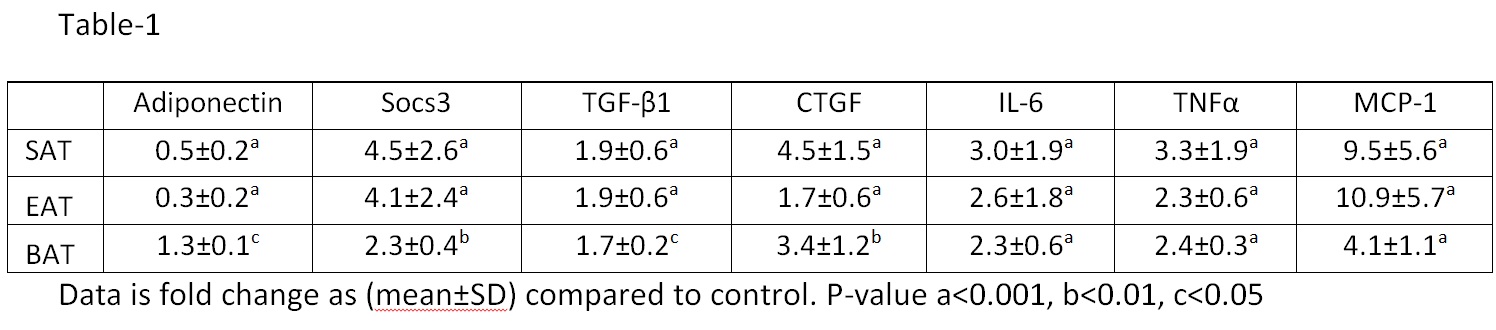
Fat tissues were collected from male mice after 30 weeks of a high fat diet (HFD:45% fat). Age matched mice fed normal chow were controls. HFD increased body weight (by HFD:29.6±4.2g vs C:9.3±3.3g, P<0.05), fasting blood glucose (HFD:9.4±1.2 vs C:7.6±1.2 mmol/L, P<0.05), and IR determined by ITT (HFD:1.4±0.4 vs C:1.0±0.1, P<0.05). Consistent with IR, HFD increased Socs3 and decreased adiponectin in SAT and EAT (Table-1). HFD also increased inflammatory markers (IL-6, TNFα and MCP-1) and profibrotic markers TGFβ1 and CTGF (Table-1). In HFD mice impaired adipogenesis in SAT and EAT was shown by decreased PPARγ (by 0.7±0.3 and 0.5±0.3 fold, P<0.05), PGCα was only decreased in EAT (by 0.6±0.3 fold, P<0.05). The higher BAT adiponectin (Table-1), UCP-1, PPARγ and PGCα1 (by 4.1±0.9, 1.9±0.2 and 1.6±0.3 fold, P<0.05) suggests that in this depot insulin sensitivity and adipogenesis may be maintained.
Results showed HFD decreased insulin sensitivity, impaired adipogenesis and increased fibrotic and pro-inflammatory changes in SAT and EAT. In contrast, in BAT metabolic function is preserved despite increased inflammatory and profibrotic markers. This is consistent with the proposed protective effect of BAT with improved adiponectin and increased UCP-1.